HOW CAN WE HELP YOU? Call 1-800-TRY-CHOP
Center for Human Phenomic Science
The Center for Human Phenomic Science (CHPS) provides the resources, environment, operations, and training to promote high-quality clinical and translational research by qualified investigators. CHPS was formed with the receipt of the Clinical and Translational Science Award (CTSA), an NIH Roadmap initiative. CHPS is a CHOP-Penn collaboration with child- and adult-specific facilities, as well as joint components, supporting research at the interface of basic and clinical research with a focus on the development of new and safer therapeutic entities.
The heart of CHPS is its Clinical Cores. The Cores are designed to facilitate the research process and are overseen by a director with extensive expertise who serves as a resource for investigators and trainees.
Interested in Working with CHPS?
To request service from CHPS, study teams must complete the eCHPS application within the CHOP eResearch protocol submission process. See our helpful how-to guide to CHPS Submissions, Amendments, and Continuing Reviews to get started with your eCHPS application.
Once the eCHPS application is completed and submitted, the request will be reviewed by the Directors of the Cores being requested and our Resource Committee.
After you submit the CHPS study, check your eCHPS workspace for any correspondence and the status of your study.
Studies must receive both IRB and CHPS approval before Core services can be utilized. In addition, all studies must be fully signed-off and in the status of released/open to accrual in OnCore before registering or scheduling subjects for CHPS visits.
If you plan to use CHPS at Penn, please reach out directly at the Penn Center for Human Phenomic Science
CHPS Clinical Cores
-
CHPS Research Nursing Core

-
CHPS Cardiovascular Core

-
CHPS Nutrition Assessment Unit
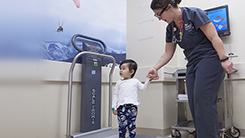
-
CHPS Nurse Practitioner Core

-
CHPS Behavioral Neuroscience Core

-
CHPS Bionutrition Unit

-
CHPS Ophthalmology Core
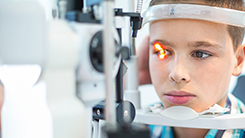
-
CHPS Sleep Core
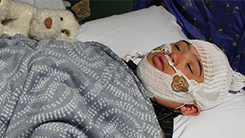
-
CHPS Informatics Core

-
CHPS Digital Health Innovation Core

-
CHPS Study Design and Biostatistics Core


Lorraine L. Katz, MD

Andrea Kelly, MD, MSCE

Babette S. Zemel, PhD
